Penny Hatzimanolakis
Department of Oral Biological and Medical Sciences, University of British Columbia, Vancouver, British Columbia, Canada.
Correspondence: Penny Hatzimanolakis, Dip. DH, BDSc, MSc, Clinical Associate Professor University of British Columbia, email: pennyhat@dentistry.ubc.ca
The Dental Implants Global Market Report 2021 had expected growth to $4.486 billion with a compound annual growth rate (CAGR) of 8.7%1,2, explaining why there are over 500 plus registered dental implant exhibitors at the fall (2021) International Dental Show (IDS). The global market expansion and the upsurge of multiple implant designs/components understandably make best practice protocols arduous for all clinicians.
Planning, placing, and restoring an implant is fundamental for success, but the implant’s management quickly shifts to the hygiene practitioner for long-term longevity. The purpose of this article is to elucidate the anatomic features of the peri-implant tissues and reconsider biofilm management to better support clinicians’ decision-making in achieving a patient-centred care approach.
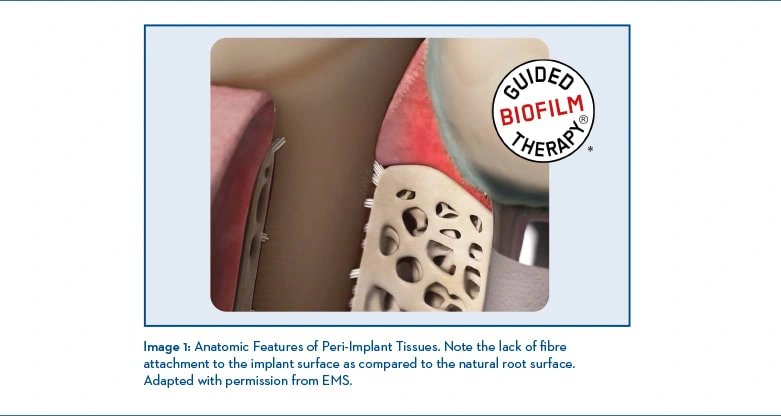
Anatomic Features of Peri-Implant Tissues Although similar to periodontal health around natural teeth, the peri-implant anatomic features and functions have significant structural differences. In particular, the involvement with the implant surrounding tissues and biological attachment.3
From Greek origin, peri meaning “around” or “enclosing” is the area around the implant/abutment which is composed of a soft tissue zone, the peri-implant mucosa, which forms during the wound healing process4, and the hard tissue zone, forming the attachment to the implant surface to achieve implant stability.4 Furthermore, the peri-implant mucosa facing the implant/abutment contains two separate sections,5 a coronal area lined by a thin non-keratinized barrier epithelium and sulcular epithelium, and an apical area in which the connective tissue seems to be in contact with the implant surface. This apical section of the peri-implant mucosa, although controversial, is designated the zone of connective tissue adhesion/leaning and can exhibit a variation between 1 and 2 mm.6 The significant differences between periodontal and peri-implant tissues are relevant in understanding the potential destructive patterns. Professional judgement in deciding an instrument (assessing or debriding) or oral self-care products for implant/abutment maintenance needs to respect the implant/abutment’s biomaterials, the peri-implant tissue biology, given its dual function in protecting the underlining bone and the bone supporting implant5, and reduce invasiveness7 to the thin lining and minimal adhesion areas.
Etiology and Microbiology of Peri-implant Tissue Infections
Periodontal disease is multifactorial, and peri-implant tissue infections/diseases are susceptible to comparable risks. An individual’s oral microbiome is either symbiotic, i.e., commensal and vital for healthy soft and hard dental tissues or dysbiotic, i.e., imbalance communities eliciting an inflammatory host response that correlates to microbe-related pathologies like peri-implant infections8. The primary etiology of peri-implant infections from a biological perspective is the shift of an ecological environment to a dysbiotic biofilm. Keystone pathogens like Porphyromonas gingivalis can disrupt all species, pathogenic or not, into a collective inflammophilic dysbiotic8 community and destructive to the peri-implant tissues. The constant exposure to microorganisms in the oral microbiome challenges the peri-implant mucosa, including the adhesion (seal). Data from Tomasi et al.9 suggest that the bacteria surrounding exposed dental implants form a diverse microbiome regardless of the periodontal profile of patients.
The Peri-Implant classification: shaping clinician-recommendations for improved patient outcomes
The inaugural AAP/EFP implant classification provides a workflow for diagnosing clinical cases.7 Peri-implant health is the absence of tissue inflammation and further additional bone loss following initial healing. In contrast, peri-mucositis presents with tissue inflammation combined with no additional bone loss following initial healing, and peri-implantitis, the advancement of disease, has an unfavourable change in probing depth, the presence of tissue inflammation combined with radiographic evidence of bone loss following initial healing.8
Peri-Implant Biofilm Management
Patients’ care plans aim to prevent, arrest or reduce the disease progression by incorporating best practices in mitigating peri-implant inflammation both professionally and essentially by daily patient-self-care regimens. The accepted periodontal disease model implies the process involves a shift to a dysbiotic biofilm, initiating inflammation and resulting in an infection. However, in a proposed model, there is evidence suggesting inflammation is the initiator, i.e., inflammation starts the process and is responsible for the ecological biofilm shift, resulting in infection not only in periodontal diseases but in other systemic inflammatory conditions.10 Although mechanical biofilm disruption remains the gold standard and is achieved safely with peri-implant patients using electric toothbrushes with the oscillation-rotation mechanism and interdental devices,11 the implant/prosthetic topography can be challenging and alone may be inadequate in the prevention or management of inflammation. What about therapeutic agents in self-care products? Are they all the same? Prescribing self-care regimens, products, or particular agents must be individualized, effective, safe, non-altering to the implant surface integrity, non-degrading biomaterials, and most importantly, support the peri-implant tissues in a symbiotic (healthy) microbiome and reduce the risk of an ecological biofilm shift. Using a dentifrice with a stabilized stannous fluoride (SnF2) composition can disrupt the biofilm by reducing its metabolic production of toxins12, directly suppressing pathogen virulence factors13, with a subgingival penetration of up to 4 mm with natural teeth.14 Biesbrock A et al. demonstrated a 3.7x better odds of shifting to generally healthy (<10% bleeding sites) vs. a negative control sodium fluoride (or MFP) toothpaste using Crest’s SnF2 formulation.15 The nearly neutral pH formulation also demonstrated no corrosion on titanium-alloy in an in-vitro study and was just as safe as sodium fluoride.16 To support the critical decision-making in selecting a methodology for professional or patient self-care regimens, they should align with; 1) what is the peri-implant tissue health, 2) what is the etiology and contributing factors, 3) what methodology will support the disruption and therapeutic management of biofilm and 4) what methodology will respect the biology of the peri-implant tissues and be non-invasive.
Conclusion
Dental hygiene practitioners should be well-versed in the fundamental principles of implant designs, abutments, and the features and functions of the peri-implant tissues to correctly identify implant-related diseases. As with periodontally involved patients, new and maintenance protocols must include all risk assessments, including social determinants of health, professional guided biofilm management, effective oral self-care products and regimens, and ongoing, consistent recare, which can significantly impact implant success and survival.
*https://www.ems-dental.com/en/guided-biofilm-therapy
References:
1. https://www.reportlinker.com/p06071073/Dental-Implants-Global-Market-Report-COVID-19-Growth-And-Change-to.html?utm_source=GNW.
2. Elani HW, Starr JR, DaSilva JD, Gallucc3i GO. Trends in Dental Implant Use in the U.S., 1999-2016, and Projections to 2026. J Dent Res. 2018; 97(13):1424-1430. doi: 10.1177/0022034518792567.
3. Renvert S, Persson GR, Pirih FQ, Camargo PM. Peri-implant health, peri-implant mucositis, and peri-implantitis: Case definitions and diagnostic considerations. J Clin Periodontol. 2018;45:S278-S285.
4. Berglundh T, Abrahamsson I, Welander M, Lang NP, Lindhe J. Morphogenesis of the peri-implant mucosa: an experimental study in dogs. Clin Oral Implants Res;2007;18:1–8.
5. Araujo MG., Lindhe J. Peri-implant health. J Clin Periodontol. 2018;45:S249-S256.
6. Larjava H, Koivisto L, Hakkinen L, Heino J. Epithelial Integrins with Special Reference to Oral Epithelia. J Dent Res. 2011 Dec; 90(12): 1367–1376. doi: 10.1177/0022034511402207.
7. Saadoum AP, Touati B. Soft Tissue Recession around Implants: Is it still unavoidable? -Part II. Pract Proced Aesthet Dent. 2007;19(2):81-87.
8. Cortés-Acha B, Figueiredo R, Seminago, et al. Microbiota Analysis of Biofilms on Experimental Abutments Mimicking Dental Implants: An In Vivo Model. J Periodontol. Oct 2017; 88(10):1090-1104.
9. Tomasi C, Tessarolo F, Caola I, et al. Early healing of peri-implant mucosa in man. J Clin Periodontol. 2016;43:816–82.
10. Van Dyke TE. Shifting the paradigm form inhibitors of inflammation to resolvers of inflammation in periodontitis. J Periodontol. Oct 2020; volume 91 (Number S1) pgs: S19.
11. Vandekerckhove, B., Quirynen, M., Warren, P.R. et al. The safety and efficacy of a powered toothbrush on soft tissues in patients with implant-supported fixed prostheses. Clin Oral Invest 8, 206–210 (2004).
12. 4 Cannon M, Khambe D, Klukow-ska M, Ramsey D, Miner M, Huggins T, White DJ. Clinical effects of stabilized stannous fluoride dentifrice in reducing plaque microbial virulence II: Metabonomic changes. J Clin Dent 2018;29:1–12.
13. Klukowska M, Haught JC, Xie S, Circello B, Tansky CS, Khambe D, Huggins T, White DJ. Clinical effects of stabilized stannous fluoride dentifrice in reducing plaque microbial virulence I: Microbiological and receptor cell findings. J Clin Dent 2017;28:16-26.
14. Tao et al. Assessment of the Effects of a Novel Stabilized Stannous Fluoride Dentifrice on Gingivitis in a Two-Month Positive-Controlled Clinical Study J Clin Dent. 2017 Dec;28(4 Spec No B): B12-16.
15. Biesbrocck A, et al. The effects of bioavailability gluconate chelated stannous fluoride dentifrice on gingival bleeding: Meta-Analysis of eighteen randomized controlled trials. J Clin Periodontol (2019) December.
16. Suszcynsky-Meister E et al. J Dent Res (2018) 97 (Spec Iss A): Abstract 994.